HPLC Basics
HPLC basic information
HPLC basics information on liquid chromatography in general and HPLC in particular.
- 1.0 The HPLC
- 2.0 Types of Liquid Chromatography
- 3.0 HPLC Versions
- 4.0 HPLC Systems
- 5.0 HPLC Pumps
- 6.0 Sample Application
- 7.0 HPLC Columns
- 8.0 HPLC Detectors
- 9.0 Fraction Collection
- 10.0 HPLC Software
The term solid/liquid chromatography summarizes physical methods which lead to substance separation by distribution between a stationary solid phase, and a moving, mobile, liquid phase (e.g. HPLC). Necessary prerequisites are that the mixtures to be analyzed are present dissolved in a solvent and that the substances show low volatility or are non volatile.
For volatile substances mostly gas chromatography is used. The term liquid/liquid chromatography summarizes physical methods which lead to substance separation by distribution between a stationary liquid phase and a moving,mobile liquid phase (see CPC or distribution chromatography).
1.0 The HPLC
In the 1960s the column chromatography LC with its low-pressure suitable glass columns (Fig. 1) was further developed to the HPLC with its high-pressure adapted metal columns (Fig. 2).
- Fig 1
- Fig 2


![]()
![]()
HPLC = High Performance Liquid Chromatography
LC = Liquid Chromatography
The HPLC, just as the LC, is used for substance separation from a sample, also known as purification or isolation, however, the separation via HPLC is completed a lot faster than with LC. A prerequisite for liquid chromatography is that the mixtures to be analyzed are present dissolved in a solvent.
The purification takes place in a separation column between a stationary and a mobile phase. In this type of chromatography, the stationary phase (Fig. 3) is a granular material with very small porous particles in a separation column. The mobile phase, on the other hand, is a solvent or solvent mixture which is forced at high pressure through the separation column. Via a valve with a connected sample loop (Fig. 4), i.e. a small tube or a capillary made of stainless steel, the sample is injected into the mobile phase flow from the pump to the separation column using a syringe.
- Fig 3
- Fig 4





Mobile Phase
Also Known as Eluent, Eluant or Flow Agent
Subsequently the individual components of the sample migrate through the column at different rates because they are retained to a varying degree by interactions with the stationary phase. After leaving the column, the individual substances are detected by a suitable detector and passed on as a signal to the HPLC software in the computer. At the end of this operation/run a chromatogram in the HPLC software on the computer is obtained (Fig. 5). The chromatogram allows the identification and quantification of the different substances.
- Fig 5


The HPLC has developed into a universally applicable method, so that it finds its use in almost all areas of chemistry, biochemistry and pharmacy.
Typical applications of HPLC
- Analysis of drugs
- Analysis of synthetic polymers
- Analysis of pollutants in environmental analytics
- Determination of drugs in biological matrices
- Isolation of valuable products
- Product purity and quality control of industrial products and fine chemicals
- Separation and purification of biopolymers such as enzymes or nucleic acids
2.0 Types of Liquid Chromatography
2.1 Adsorption Chromatography
With silica gel or alumina as stationary phase. The weak interaction bond between a molecule and the silica gel is called adsorption, the dissolution of the bond desorption. Because the molecules of the eluent occupy the active sites of the silica gel prior to the molecules of the sample, the sample molecules can only be adsorbed when their interaction with the active sites is stronger than that of the eluent molecules. The sample molecules are bound by dipole-dipole interactions reversibly to the stationary phase. The retention time of a substance by the stationary phase varies depending on the different degrees of interaction with the surface of the stationary phase. This is the mechanism that separates the sample constituents.
2.2 Affinity Chromatography
Uses the tendency of substances to react with one another as a chromatographic method. Of the two components which show affinity, one is the ligand, which is firmly bound to the carrier, and the other is the sample, which is adsorbed from the eluent. Substances that do not fit to the ligand, are not adsorbed and are washed away by the eluent towards the column outlet.
2.3 Gel Permeation Chromatography
Differs completely from the other chromatography methods. Here, the eluent, mobile phase, does not interact with the stationary phase. The substance separation of a sample is obtained by penetration of the sample molecules into the interspaces of the stationary phase. Too large molecules are washed away with the mobile phase to the column outlet. The smaller molecules thus reach the column exit later.
In normal phase distribution chromatography, the stationary phase is more polar than the mobile phase; in reverse-phase chromatography (RP-Reverse Phase) the mobile phase is more polar than the stationary phase. The stationary phase can be chemically bound to a carrier material or the carrier material is simply coated with the stationary phase. The reverse-phase chromatography is mainly used for polar or nonpolar substances.
2.4 Distribution Chromatography
Also known as distribution chromatography is a novel method of liquid chromatography. It resembles a separation method, in which the sample is transported under pressure by a liquid mobile phase through the liquid stationary phase in the inner chambers (Fig. 6) of a rotor (Fig. 7). The analyte that is better soluble in the stationary phase is retained longer, while the analyte that is better soluble in the mobile phase is transported faster with the liquid mobile phase.
- Fig 6
- Fig 7




3.0 HPLC Versions
A distinction is made primarily between isocratic HPLC and gradient HPLC.
3.1 Isocratic HPLC
Is carried out with only one eluent composition within a single run. Therefore, the interaction between mobile and stationary phases is constant during the entire time span of the separation. Especially similar substances can be separated well with this method. This type of implementation is particularly popular especially in routine applications and should be as far as possible strived for all separation tasks. Another advantage of this method is that you only need a single pump for pumping the eluents.
3.2 Gradient HPLC
The composition of the eluent is changed continuously over time. A constant change in interaction between eluent and stationary phase is achieved this way. The sample substances are displaced quicker from the exchange sites on the stationary phase. This renders the separability of components with very different polarity in acceptable time possible. Since eluent composition at the beginning of this separation procedure differs from that in the end, make sure the system is in equilibrium before starting the separation (equilibration). The mixing of the eluent can be carried out differently depending on the technically circumstances of each device.
3.2.1 Low Pressure Gradients
The mixing of the differnt eluents is performed by a proportioning valve before entering the pump head and the mixture is transported by the pump through the plant. This is known as a low-pressure gradient. The mixing is accomplished at the low pressure side (atmospheric pressure) of the plant, before entering the pump.
3.2.2 High Pressure Gradients
Each eluent is pumped by a separate pump and the mixing is carried out on the pressure side of the system. For this purpose, a mixing chamber is used. Such a system is referred to as high-pressure gradient HPLC. The transport of the eluent through the HPLC system builds up pressure. The major portion in this case comes from the column. Depending on the separation material, its granulation and shape, the column length and the used eluent (different viscosity) pressures up to 300 bar and more can build up.
4.0 Composition of Selected HPLC Systems
A standard HPLC consists of at least five main devices: pump, injection unit, separation column, detector with evaluation software and fraction collector. For HPLC separation particles with particle sizes from 3 to 20 µm are used. Large separation plate numbers are realizable on the one hand, on the other the mobile phase has to overcome relatively high back pressure during transport through the separation column. All units have to be pressure resistant and connected preferably dead volume free to one another.


The analytical HPLC usually consists of one HPLC pump with a flow range of 0.01 – 10 ml/min, one UV detector, one sample injector (autosampler) and a PC with analytical HPLC software.


The semi preparative HPLC usually consists of one HPLC pump with a flow range from 0.01 – 50 ml/min, one separation column, one UV detector, a fraction collector and a PC with preparative HPLC software.


The preparative HPLC consists mostly of two HPLC pumps with a flow range from 0.1 – 100 ml/min, one separation column, one UV-Vis detector, an autosampler/fraction collector and a PC with preparative HPLC software.


The HPLC for production consists mostly of two HPLC pumps with a flow range of each > 1000 ml/min, one sample injection pump, one column packing station, one UV-Vis detector, a fraction collector and a PC with preparative HPLC software.


The system for liquid-liquid extraction, or distribution chromatography consists mostly of one/two HPLC pump/s, a SCPC with its rotary separation column, one UV-Vis detector, a fraction collector and a PC with HPLC software.
5.0 HPLC Pumps
5.1 The HPLC piston pump
Is an electro mechanical device for transport of the mobile phase (eluent, flow medium) at high pressure up to about 600 bar. Inside the pump head a piston is moved by seals back and forth, whereby the eluent is sucked through the inlet valve into the piston chamber and forced through the outlet valve toward the separation column (Fig.8). By using several pump heads a pulsation free eluent flow can be ensured. While one of the pistons is sucking the other piston releases the previously sucked up eluent via the outlet.
- Fig 8


5.1 Characteristic for the HPLC diaphragm pump
Is that the eluent is not transported by piston as is true for the piston pump, but rather by the movement of a diaphragm. The membrane is put into motion by the to and fro movement of an oil surrounded piston whereby the eluent is sucked and ejected. Ball valves also regulate the liquid flow path here. This type of pump is mainly used in industrial preparative HPLC systems.
6.0 Sample Application
The sample application can be done manually with a syringe via a valve with sample loop (Fig. 9) or automatically via an autosampler (Fig. 10). The sample has to be injected under ambient pressure into the pressurized line of the plant.
- Fig 9
- Fig 10





6.1 For manual sample loading
The sample is drawn up into a syringe and firstly injected without pressure into the sample loop, which is located on a multi-way valve. By subsequently switching the valve to the pressure side the eluent flow passes through the sample loop towards the separation column.
Some autosamplers offer possibilities for sample preparation. These include automated dilution and mixing of the sample and the pre-column derivatization. With the appropriate HPLC software the devices dilute highly concentrated samples automatically, so that after only a few injections the highest analysis accuracy is achieved and column overload is prevented.
Should the concentration lie outside the calibrated range, these over-range samples are diluted as well making further analyzing unnecessary. To shorten analysis time, some devices work with the sample overlap function. While one sample is being separated the following is already being prepared. Mixing with an internal standard, the addition of reagents or the production of standard solutions and calibrations are other possible functions of the autosampler.
7.0 HPLC Columns, HPLC Separation Columns
HPLC columns usually consist of a stainless steel tube and screw caps on both ends with connections for capillaries as an inlet on one side and as an outlet on the other side. The HPLC columns are filled without spacing under mechanical pressure with the stationary phase, a very fine-grained, porous powder (3, 5, 8 or 10 µm particle size). The smaller the grain size, the higher the separation efficiency. The column material is always selected according to the separation task. Probably the most commonly used HPLC column is the “C18-column”.


The stationary phase is in this case a chemically modified silica gel with built up C18 chains on the surface. In order to ensure a good reproducibility of the results the HPLC column should be operated at a constant temperature. Inside the HPLC column the sample substances are adsorbed onto the stationary phase and separated by adsorption/desorption processes.
Analytical separation columns mostly have an inner diameter of 2 to 5 mm and a length of up to 250 mm, they are designed for a max. flow of about 2 ml/min.
Semi preparative separation columns with an internal diameter of 10 – 50 mm and a length of 250 mm are designed for a max. flow of about 100 ml/min.
Preparative separation columns with an inner diameter of 75 mm and a length of up to 1000 mm are used for flow rates of more than 100 ml/min.
The pre column is installed directly before of the separation column to prevent it from clogging. The pre column is smaller and almost always has the same column material as the separation column.
7.1 The Column Packing Station “Packing Columns Yourself”
If you want to be flexible with the column material in your separation column, use a column packing stand also known as column packing station.
Using static or dynamic axial compression all current column materials can be packed reproducibly in the desired bed length by the user.


The simple packing process allows quick adaption of the HPLC column to recent requirements making a selection of expensive pre-packed columns unnecessary. The carrier material can be packed with high pressures as a frit is firmly connected to the moving piston. This ensures uniform distribution of the substance over the entire column cross-section and an optimal shape of the peaks.
The compression of the column bed can be achieved in two ways, either with a space-saving manual hydraulic or with a pneumatically driven hydraulic for static and dynamic axial compression. In case of static compression the HPLC column can be taken off the packing station. This way multiple HPLC columns can be prepared with a single packing station.
In case of dynamic axial compression, however, the HPLC column remains in the packing station and the column bed is automatically maintained at pressure, preventing spacing at the column inlet which could affect the separation efficiency. Decreasing separation efficiency of the withdrawn HPLC column due to occurred spacing is reversed easily by repressing the column material.
Furthermore, varying bed lengths can be realized with a single column tube using a column packing station and as an option a column tube with thermal jacket is available making temperature control over the entire bed length possible.
The Packing Process


The bottom flange is mounted on the column tube. The slurry (emulsion of carrier material and suitable solvent such as isopropyl alcohol) is filled into the column tube. The slurry (column material) is pressed hydraulically; the slurry solvent is displaced from the column tube forming a homogeneous solid packing from the packing material. The piston is fixed (static compression) or automatically pressed (dynamic axial compression).
7.2 Rotary Column
This device with over 1000 serially interconnected chambers (Fig. 7) inside a rotor (Fig. 6) is called SCPC. The sample components are separated and fractionated after passing through those chambers. This separation method is referred to as distribution chromatography, liquid-liquid extraction, Centrifugal Partition Chromatography (CPC) or Counter Current Chromatography (CCC). The separation process takes place in two immiscible liquid phases, whereby the components of the sample are distributed to either of the two liquid phases according to their respective polarity.
This method allows separations of samples of various kinds in analytical, preparative and productional scale. The schematic construction of the SCPC systems is always identical regardless of size and flow rate. The heart of each system is the rotary column, the rotor. It resembles the separation column surrounded by peripheral devices, such as HPLC pump, sample injection system, detector, fraction collector and computer with HPLC software for system control and chromatogram evaluation.
Compared to HPLC, the SCPC system runs with fewer problems, is simpler to handle and less expensive, especially if crude extracts of different matrices need to be separated. SCPC systems do not directly compete with HPLC, but rather accelerate the entire chromatographic process to the pure product in combination with the latter.
8.0 HPLC Detectors
HPLC detectors have the task to record the separated sample molecules diluted in the mobile phase after their separation on the separation column and to transfer this information to the HPLC software in the computer.
8.1 The UV detector
Is currently the most frequently used detector in the HPLC. UV-light with a specific wavelength from a deuterium or quartz lamp in the detector passes through the flow cell. Followed by detection via a photodiode (Fig. 14) it is passed on as an electric signal to the HPLC software. The UV detector constantly measures the light extinction after transmission through the eluent with the sample molecules, flowing through two glass plates with defined constant distance, via a photo meter cell in the flow cell.
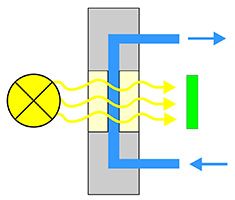

The attenuation of the radiation (extinction) and thus the change of the electrical signal, therefore, only correlates with the concentration and type of medium. Since eluent and sample absorb the light differently this results in a varying electrical signal. These changes appear as substance peak in the HPLC software. The chromatogram mostly is composed of several peaks.
Detection threshold about 0.3 ng/ml.
8.2 The diode array detector (DAD)


Or also referred to as photodiode array detector (PDA), measures, just as the UV-detector, light absorption by the mobile phase in the ultraviolet and visual wavelength range. However, the conversion to an electrical measurement signal does not occur via a photo cell, but rather by 512 or 1024 photodiodes arranged in a square, the diode array. This makes it possible to detect the entire spectral range, i.e. to detect multiple wavelengths simultaneously.
Detection threshold about 0.3 ng/ml.
8.3 Refractive Index detector (RI)
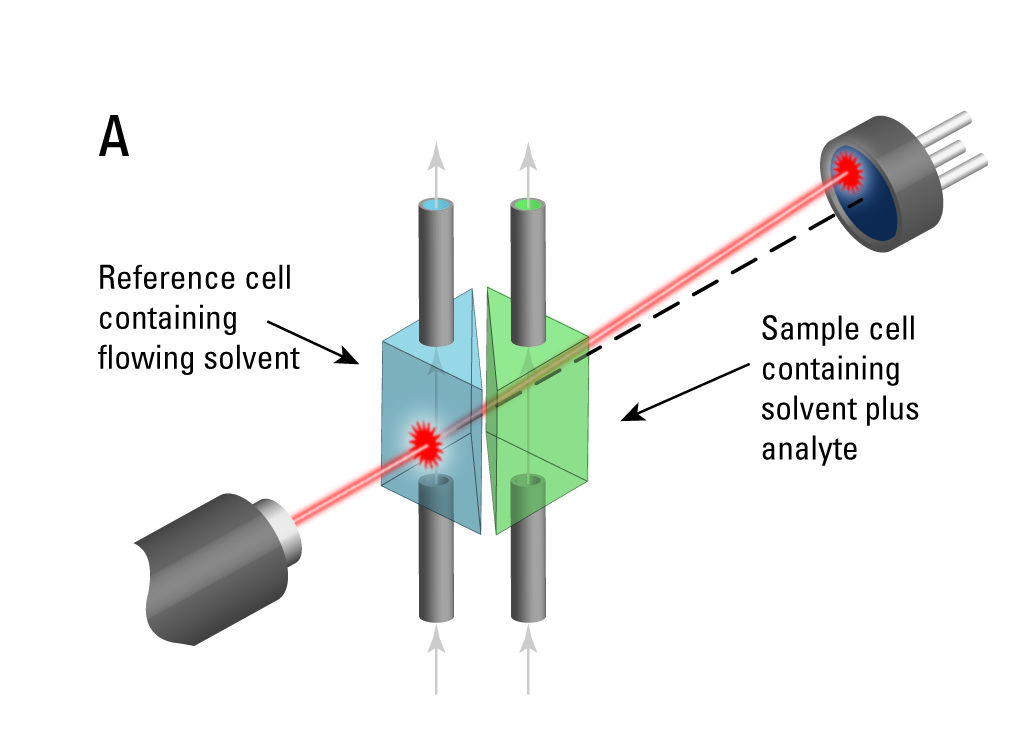

Is less sensitive than the UV-detector, but it can detect substances that show no UV absorbance. It measures the difference of the refractive index between pure eluent and eluent mixed with the sample. For detection the RI detector, however, always needs a reference channel with pure mobile phase. The more the refractive indices of the mobile phase and the reference substance differ, the greater the signal for the substance peak in the chromatogram. This detector is mainly used in polymer chemistry and in sugar analytics.
Detection threshold about 0.7 µg/ml.
8.4 Fluorescence Detector (FLD)
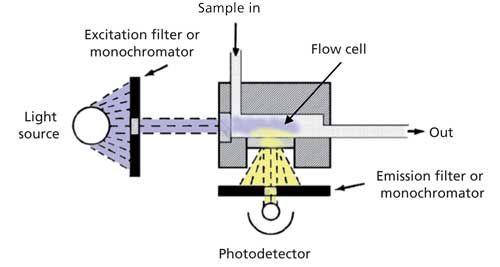

Is up to 1000 times more sensitive than the UV detector. It uses the property of fluorescent substances to detect those.
Detection threshold about 0.8 pg/ml.
8.5 Electrochemical detector (ECD)


Oxidizes or reduces substance components by applying a potential. The change of potential indicates the presence of a substance component. Its field of application is mainly clinical chemistry.
Detection threshold about 1.0 pg/ml
8.6 Evaporative Light Scattering Detector (ELSD)


Is used when substance classes such as lipids, carbohydrates, polymers, surfactants, amino acids are not or only slightly UV or VIS-active. In the HPLC and GPC it is a very good universal detector, because it uses a physically different detection principle. The eluent is nebulized with compressed air/nitrogen and vaporized in a drying unit made of glass. In the resulting aerosol particles the analytes are measured mass selectively via light scattering. In contrast to the RI detector and electrochemical detector (ECD) this detection principle allows the application of eluent gradients. The light scattering technique ensures highest sensitivity and drift-free detection without complicated setup procedures.
Detection threshold about 5 ng/sample component.
9.0 Fraction Collector
A fraction collector ensures you don’t lose valuable substances after a successful separation. Besides drop and volume dependant collection especially fractionating over time periods, timeframe and/or peak recognition is very helpful. Optional valves guarantee contamination-free collection in the individual vessels.


Various rack formats from microtiter plates to a large volume scale usually allow a wide range of applications. Mostly programs can be stored and recalled automatically by the HPLC software. Via an integrated sample tracking the software allows an accurate classification of your fractions to the recorded collection events in the chromatograms as well as the documentation of the fractionation.
10.0 HPLC Software
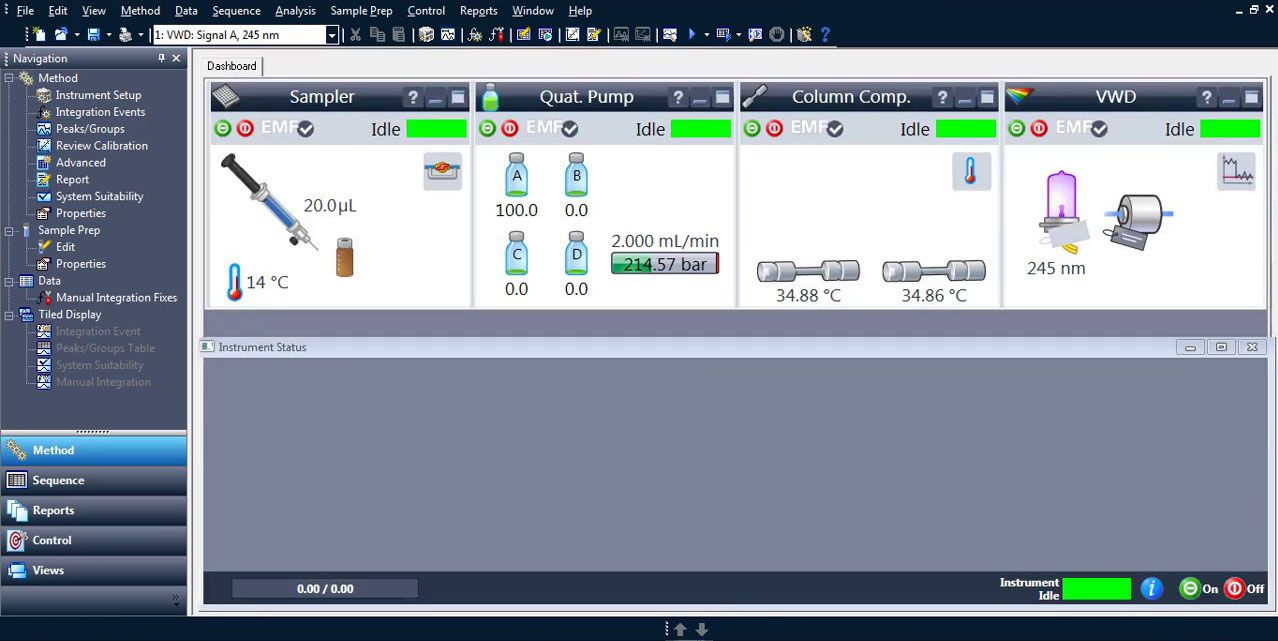

A computer with HPLC software is mostly used for controlling the system and evaluating the measurements. For qualitative analysis only those peaks included in your sample are separated (fractionation). For quantitative analysis the exact concentration or amount of substances present in the sample must be known. Hence the evaluation over the area of the peaks is possible.
10.1 The Chromatogram


A computer with HPLC software is mostly used for controlling the system and evaluating the measurements. For qualitative analysis only those peaks included in your sample are separated (fractionation). For quantitative analysis the exact concentration or amount of substances present in the sample must be known. Hence the evaluation over the area of the peaks is possible.
